Fetal Lower Urinary Tract Obstruction (LUTO)
LUTO is a rare condition where the baby’s urine flow is blocked inside the womb. This causes the baby’s bladder to swell and get very large. Because the baby cannot urinate normally, the amount of amniotic fluid (the fluid around the baby) decreases. Since after the middle of pregnancy amniotic fluid mainly comes from the baby’s urine, low fluid can cause serious problems.
The urine blockage puts pressure on the baby’s urinary system and kidneys, which can cause kidney damage (called renal dysplasia). Low amniotic fluid also affects lung development, potentially leading to lung problems (pulmonary hypoplasia). Both kidney and lung problems increase risks for the baby.
In boys, the most common cause is called posterior urethral valves (a blockage in the urinary tract). In girls, urethral atresia (where the urinary opening is missing or blocked) is most common. Other causes exist, but ultrasound often shows similar signs.
How is it diagnosed?
Ultrasound detects a large, swollen bladder and checks for kidney swelling and fluid levels. Tests on the baby’s blood (via umbilical cord) or urine (via bladder sampling) help assess kidney function. These tests guide prognosis and treatment decisions.
What are the chances and risks?
- If untreated and fluid remains low, perinatal mortality (death around birth) is about 77%.
- Treatment aims to improve these outcomes but some risks remain.
Treatment options
- Expectant management (watch and wait):
Frequent ultrasounds monitor the baby. After birth, specialists treat any kidney or bladder issues. However, low fluid increases the risk of kidney damage and lung problems. - Vesicoamniotic shunt (fetal surgery):
A small tube is placed from the baby’s bladder to the amniotic fluid to bypass the blockage. This can help protect the kidneys and improve fluid levels for better lung growth.
- About 66% of babies treated this way survive the perinatal period.
- However, half of these survivors may still have significant kidney damage needing dialysis or transplant later.
- Risks of the shunt include blockage or moving out of place (25% chance), sometimes requiring replacement, and a 4% risk of fetal death.
- About 66% of babies treated this way survive the perinatal period.
- Pregnancy termination:
This is an option before 24 weeks in some regions but is not offered here.
Who can get fetal treatment?
- Babies between 16 and 30 weeks with LUTO and good kidney function on testing are candidates.
- Babies with major birth defects, poor kidney tests, ruptured membranes, infection, or labor are not eligible.
The procedure
The shunt placement is done under local anesthesia for the mother and medicine to relax the baby. Ultrasound guides the placement of the tube to restore urine flow.
Aftercare
Frequent ultrasounds monitor amniotic fluid, bladder size, kidneys, and shunt position—weekly for the first month, then every 3-4 weeks. After birth, pediatric specialists manage ongoing care.
Fetal lower urinary tract obstruction (LUTO) is a rare condition that is caused by a blockage of fetal urination. Because the baby cannot empty the bladder, the baby’s bladder subsequently becomes very large and distended. Also, because the amniotic fluid is essentially composed of the baby’s urine beyond the middle of the second trimester, the bag of waters dries up. A cascade of secondary effects result in significant morbidity and/or mortality for the baby. This includes problems to the urinary collection system (hydronephrosis) and kidneys (renal dysplasia) attributed to the backpressure from the urinary blockage. Underdevelopment of the lungs (pulmonary hypoplasia) develops from the lack of amniotic fluid during a critical time of the pregnancy.
The cause of fetal LUTO is varied. The most common cause in male fetuses is posterior urethral valves. Oligohydramnios (low amniotic fluid volume defined as the maximum vertical pocket less than or equal to 2.0 centimeters) may then develop, and is associated with a worse prognosis. In females the most common cause is urethral atresia. Other causes of fetal LUTO include but are not limited to obstructive ureterocele, urethral stricture or agenesis, persistent chloaca, and megalourethra. The ultrasound findings of many of these conditions are similar, and it is often difficult to differentiate the cause of the urinary obstruction until after delivery.
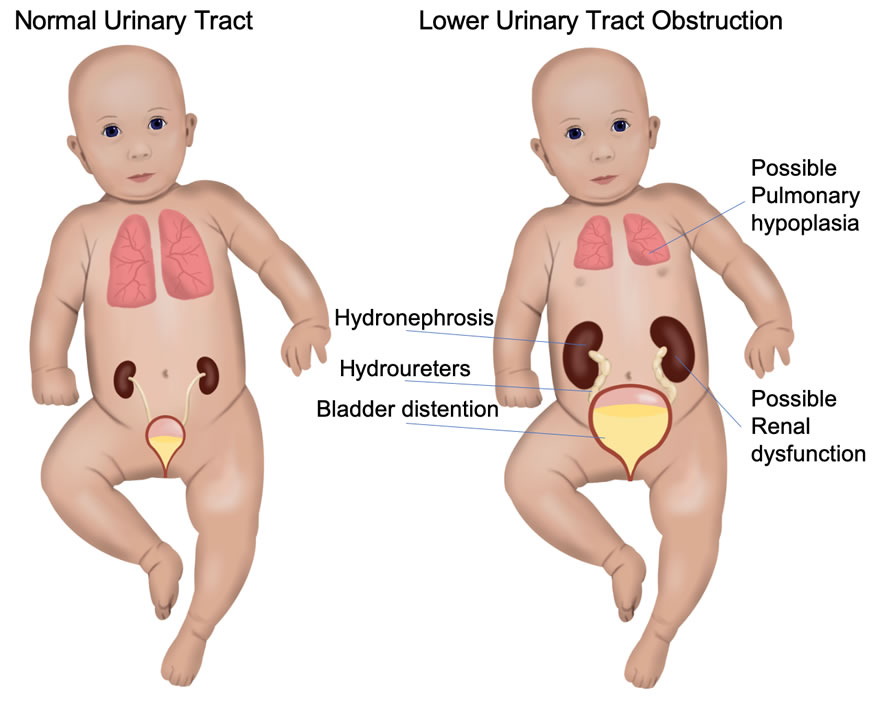
Because there are different causes of LUTO, the prognosis can be expected to be different depending on the individual diagnosis. However, a major component that dictates perinatal outcome is the secondary complications of the obstruction (renal dysplasia and pulmonary hypoplasia). To prevent these complications, several methods have been developed to bypass the blockage of urine while the baby is still in the womb, with the hope that the backpressure on the kidneys can be averted and the amniotic fluid volume may replenish to allow for more normal lung development.
Diagnosis and Prognosis Criteria
The diagnosis of LUTO is made by prenatal targeted ultrasound. Typically, the baby’s bladder is very distended. The presence of a “key-hole” sign is suggestive of posterior urethral valves, particularly in a male fetus. There may be variable degrees of dilation of the upper urinary collection system. The ultrasound findings of the baby’s kidneys should be carefully assessed for evidence of damage. The details of these ultrasound assessments are beyond the scope of this review. Assessment of amniotic fluid volume as well as the presence of other potential structural abnormalities is sought.
Once the diagnosis of LUTO is established, the prognosis for survival is then assessed. The baby’s outcomes have been correlated to the kidney function as assessed prior to treatment. There are two methods to determine the prognosis before surgery. These methods are called fetal cordocentesis, which samples the baby’s blood, and vesicocentesis which samples the baby’s urine. Genetic studies are also performed.
1. Cordocentesis (preferred): Under ultrasound guidance, a needle is placed in the umbilical cord. Fetal blood is drawn and sent for a serum Beta-2-microglobulin level. If the serum Beta-2-microglobulin level is less than 5.6 then fetal therapy may be offered.
2. Fetal Vesicocentesis: An alternative to cordocentesis is to perform a vesicocentesis. Fetal kidney function may be obtained by performing sampling of the baby’s urine by placing a thin needle into the baby’s bladder and draining the urine. If the results of the first drainage are below the threshold values (see below) then fetal therapy may be offered. If the first vesicocentesis shows values above the threshold, a repeat vesicocentesis will be performed in 48 hours. The fetal urinary parameters are as follows:
- Sodium 100 mEq/dl
- Chloride 90 mEq/dl
- Calcium 8 mEq/dl
- Beta-2-microglobulin 10 mg/L
- Osmolality 210 mOsm/L
- Total protein 20 mg/dL
Management Options and Outcomes
As mentioned above, our ability to evaluate kidney function is somewhat imprecise. This probably reflects the different diagnoses responsible for the sonographic findings, which do not have a similar prognosis despite comparable fetal urinary findings or vice versa. Thus, discussion of managements options and outcomes is somewhat hampered by this limitation. Despite this limitation, the follow treatment options are available:
1. Expectant Management: This approach entails frequent ultrasound assessment to assess progression during the pregnancy. After delivery, pediatric specialists will evaluate the baby and subsequently offer treatments at that time. The risk of this approach is that further kidney and lung damage may occur during the pregnancy. In the setting of oligohydramnios (low amniotic fluid volume), the expected perinatal mortality rate is 77%.
2) Vesicoamniotic Shunt: Fetal urinary diversion procedures have been performed since the 1980’s. Essentially, a shunt is placed between the baby’s bladder and the amniotic fluid, thus relieving the blockage. This approach is meant to prevent further kidney and lung damage. The final treatment of the obstruction is performed after the birth of the child. In cases that have been determined to be in the favorable prognostic category (see above), the approximate perinatal survival using this treatment approach is 66%, of which half will have significant kidney damage and may need dialysis or kidney transplant. Risks of shunt placement include dislodgement/blockage/malfunction (25%) thus requiring multiple shunt placements during the pregnancy, urinary ascites (leakage of urine from the bladder into the baby’s abdomen), and fetal death (4%).
3) Pregnancy Termination: Pregnancy termination may be chosen as an option up to 24 weeks gestation in California. We do not offer this procedure.
Who is a candidate for fetal treatment?
The following criteria must be met to offer fetal therapy for LUTO:
Inclusion criteria
- Gestational age: 16 weeks 0 days to 30 weeks 0 days.
- Sonographic evidence of LUTO.
- Fetal renal values below threshold shown in items 5 and 6 listed below.
- Twin gestation may be included.
Exclusion criteria
- Patients unable or unwilling to participate in the study or to be followed up.
- Presence of major congenital anomalies that may affect the overall prognosis separate from LUTO.
- Unbalanced chromosomal complement.
- Sonographic evidence of renal cystic dysplasia.
- Abnormal fetal urinary parameters (>2 abnormal values).
a) Sodium >100 mEq/dl
b) Chloride >90 mEq/dl
c) Calcium >8 mEq/dl
d) Beta-2-microglobulin >10 mg/L
e) Osmolality >210 mOsm/L
f) Total protein >20 mg/dL - Abnormal fetal serum Beta-2-microglobulin ≥ 5.6.
- Ruptured membranes.
- Chorioamnionitis.
- Placental abruption
- Active labor
- The Principal Investigator finds the subject inappropriate for the study.
- Otherwise eligible, but unable to make financial arrangements.
- Maternal age <18 years.
Procedure Details
The procedure(s) will be performed under local anesthesia. Fetal anesthesia will be delivered to the fetus with a fine needle under ultrasound guidance prior to entering the fetal bladder.
Techniques
Vesicoamniotic catheter placement: A vesicoamniotic shunt will be placed in all cases over a wire guide. This can be done under ultrasound guidance. Endoscopic documentation of adequate shunt placement may be performed as well. Vesicoamniotic shunting may be necessary several times during the pregnancy due to dislodgement of the shunt or continued urinary outlet obstruction.
Postoperative Care
Before birth, it is recommended that ultrasound assessment occur every week for the first four weeks, then every three to four weeks thereafter. Ultrasound parameters of particular importance include the amount of amniotic fluid volume, measurement of the fetal bladder, assessment of the fetal kidneys and urinary collection system, presence of urinary ascites, and location of the vesicoamniotic shunt.
After birth, the child will be evaluated by pediatric specialist and may require further tests and treatments.
For further reading, please see the link below:
